How do explosions occur? What does it take? How to assess and ultimately avoid the danger? These questions arise wherever explosive mixtures can arise, for example, due to the presence of dusts.
An explosion can occur when a flammable substance in sufficient concentration comes into contact with an oxidizing agent (e.g. air) in the presence of an ignition source. The risk of a dust explosion should not be underestimated, especially in industrial areas where fine dusts such as wood, metal or organic dusts are produced.
The assessment and prevention of explosion hazards is essentially based on knowledge of the explosion characteristics, which describe the properties of a substance with regard to its explosiveness. These key figures include the lower explosion limit (LEL), the upper explosion limit (UEL), the ignition temperature, the minimum ignition energy and the maximum explosion pressure.
How do explosions occur?
When dealing with the topic of ATEX, the same terms are used again and again. To get an overview of this topic, these terms should be clarified.
A brief overview of the terms and their meaning:
- ATEX: ATmosphères EXplosibles = explosive atmospheres
- Explosive substances: solid, liquid, pasty, gelatinous substances or mixtures that can be explosive without the oxygen in the air.
- Explosive mixture/explosive atmosphere: mixture of flammable gases, vapors, mists, or swirled dusts with air or other oxidant.
- Explosion: Abrupt increase in pressure and/or temperature
Important explosion indicators on the subject of ATEX
The safety characteristics describe the fire and explosion properties of a substance. They depend on the environmental conditions, the determination method as well as the substance itself.
With the explosion indicators, suitable protective measures and necessary protective devices can be calculated and selected to suit the intended use. Furthermore, these key figures are the basis for the creation of risk assessments (explosion protection document) or also for the selection of Ex zones.
The most important key figures for dust can be found below as a practical overview:
Explosionskennzahl | Bedeutung |
|---|---|
| Brennzahl BZ | Kenngröße für das Brandverhalten von Feststoffen. Erfahrungsgemäß nimmt die Brandausbreitung bei Stoffen, die bei Raumtemperatur nur ein schwaches Abbrandverhalten zeigen, mit steigender Temperatur zu, d. h. die Brennzahl wird höher. 1 -> kein Anbrennen, z. B. Salz 2 -> kurzes Anbrennen, z. B. Malz 3 -> längeres örtliches Glimmen/Brennen, z.B. Lactose 4 -> Ausbreitung eines Glimmbrands, z. B. Tabak 5 -> Ausbreitung eines offenen Brands, z. B. Schwefel 6 -> verpuffungsartiges Abbrennen, z. B Schwarzpulver |
| Glimmtemperatur [°C] | Kenngröße für die Zündtemperatur einer Staubschicht: Unter Versuchsbedingungen ermittelte niedrigste Temperatur einer heißen Oberfläche, bei der sich eine Staubschicht von 5 mm Dicke entzündet. Mit Zunahme der Schichtdicke des Staubs sinkt die Glimmtemperatur. |
| Explosionsdruck Pmax [bar] | Höchster Druckwert eines Staub-/Luftgemisches in einem geschlossenen Behälter bei einer Explosion. |
| Explosionsgruppe III | Einteilung von Stäuben anhand ihrer spezifischen Zündfähigkeit, Unterscheidung nach Eigenschaften: IIIA (brennbare Flusen), z.B. Flock IIIB (nicht leitfähiger Staub), z.B. Pulverlack IIIC (leitfähiger Staub), z.B. Metallstaub |
| Mindestzündenergie MZE [mJ] | Unter vorgeschriebenen Versuchsbedingungen (atmosphärischer Druck, 20°C) ermittelte, kleinste Energie, die ausreicht, die zündwilligste explosionsfähige Atmosphäre zu entzünden. (EN 13237) MZE > 10.000 mJ nicht entzündlich MZE 10 - 10.000 mJ normal entzündlich MZE 3 - 10 mJ besonders entzündlich MZE < 3 mJ extrem entzündlich |
| Explosionskonstante K/KST-Wert [bar *m/s] | Klassifizierungswert, der die Brisanz einer Verbrennung ausdrückt. Er entspricht zahlenmäßig dem Wert für die maximale Druckanstiegsgeschwindigkeit bei einer Explosion eines Staub/Luft-Gemisches in einem 1 m³-Behälter. |
| Staubexplosionsklasse | Ausdruck der Explosionsfähigkeit St 1 KST - Wert: < 200 St 2 KST - Wert: 200 - 300 St 3 KST - Wert: > 300 |
| Sauerstoffgrenzkonzentration SGK [Vol%] | Wert für die höchste Sauerstoffkonzentration in einem Gemisch aus Luft, Inertgas und Gas, bei der keine Explosion möglich ist. Wird die SGK unterschritten, ist es nicht mehr möglich, eine Explosion einzuleiten (zu wenig Sauerstoff vorhanden). |
| Selbstentzündung | Entzündung einer Staubschüttung bei allseitiger Wärmeeinwirkung und Anwesenheit von Luft nach vorangegangener Selbsterhitzung. |
| Selbstentzündungstemperatur [°C] | Temperatur, bei der bei Staub Selbstentzündung auftritt. Diese ist abhängig von Staubart, Form und Größe der Schüttung sowie Dauer der Wärmeeinwirkung. |
| Partikelgröße/Korngröße [μm] | Größe der Partikel, Angabe über den Median (50% sind größer und 50% sind kleiner als dieser Wert). Kennzahl für die Zündfähigkeit des Staubes. Mit abnehmender Korngröße steigt die Neigung der Stäube zu Explosionen, d.h. je feiner der Staub, umso leichter ist er zu entzünden, und umso heftiger verläuft die Reaktion. |
| Zündtemperatur [°C] | Unter Versuchsbedingungen ermittelte, niedrigste Temperatur einer heißen Oberfläche, bei der die Entzündungen eines brennbaren Stoffes als Staub-/Luft- oder Dampf-/Luft-Gemisch eintritt. Die Zündtemperatur einer Staubschicht (Glimmtemperatur) weicht von der Zündtemperatur einer Staubwolke ab. Der niedrigere Wert bestimmt die max. zulässige Oberflächentemperatur. |
Prerequisites for an explosion
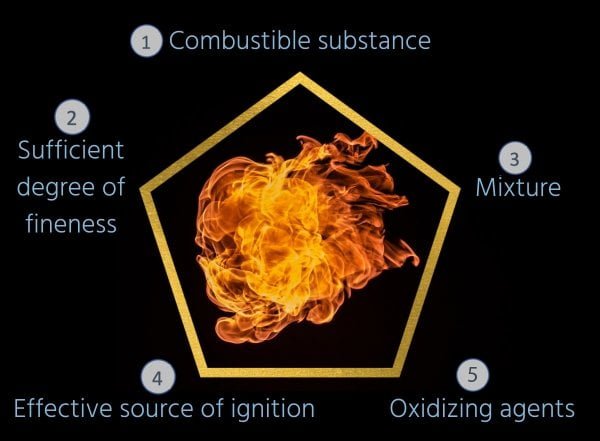
Explosions can be prevented. This requires knowledge of the “hazard pentagon”: this pentagon puts the causes of explosions in context. Here, eliminating one of these conditions for an explosion is enough to stop an explosion.
| N° | Voraussetzung | Erklärung |
|---|---|---|
| 1 | Brennbarer Stoff/ Combustible substance | z.B. in der Produktion anfallender Staub |
| 2 | Ausreichende Feinheit/ sufficient degree of finess | Je feiner der Staub, umso leichter ist dieser zu entzünden. Ist die Korngröße < 1mm besteht keine Explosionsgefahr. Je kleiner die Korngröße wird, umso wahrscheinlicher wird eine Explosion (je nach Staubart) |
| 3 | Vermischung/ mixture | Je größer die Staubdichte in der Luft, umso explosionsfähiger ist das Gemisch. Hier ist die Konzentration entscheidend: es gibt eine minimale und eine maximale Konzentrationsgrenze, die den Bereich der Explosionsfähigkeit eingrenzen (ist zu wenig oder zu viel Staub vorhanden, kann keine Explosion stattfinden). |
| 4 | Wirksame Zündquelle/ effective source of ignition | Die DIN EN 1127 beschreibt verschiedene Zündquellen. Für die Konstruktion und Anwendung von Entstaubungsanlagen bzw. Filteranlagen sind beispielsweise heiße Oberflächen, statische Elektrizität oder mechanisch erzeugte Funken zu nennen. |
| 5 | Oxidationsmittel/ Oxidizing agents | Oxidationsmittel ist z.B. der Sauerstoff in der Luft. Der Anteil kann durch Inertisierung gesenkt werden, was die Explosionsgefahr eindämmt. |
Conclusion
Depending on the type of dust, there is a greater or lesser risk of explosions occurring in a production plant. This occurrence must be avoided to protect the personnel as well as the plant and, if necessary, subsequent plants. For this purpose, knowledge of the type of dust as well as the general conditions for the formation of an explosion are of elementary importance. If it is known what will happen in the process, the risk of an explosion can be effectively eliminated in advance.


